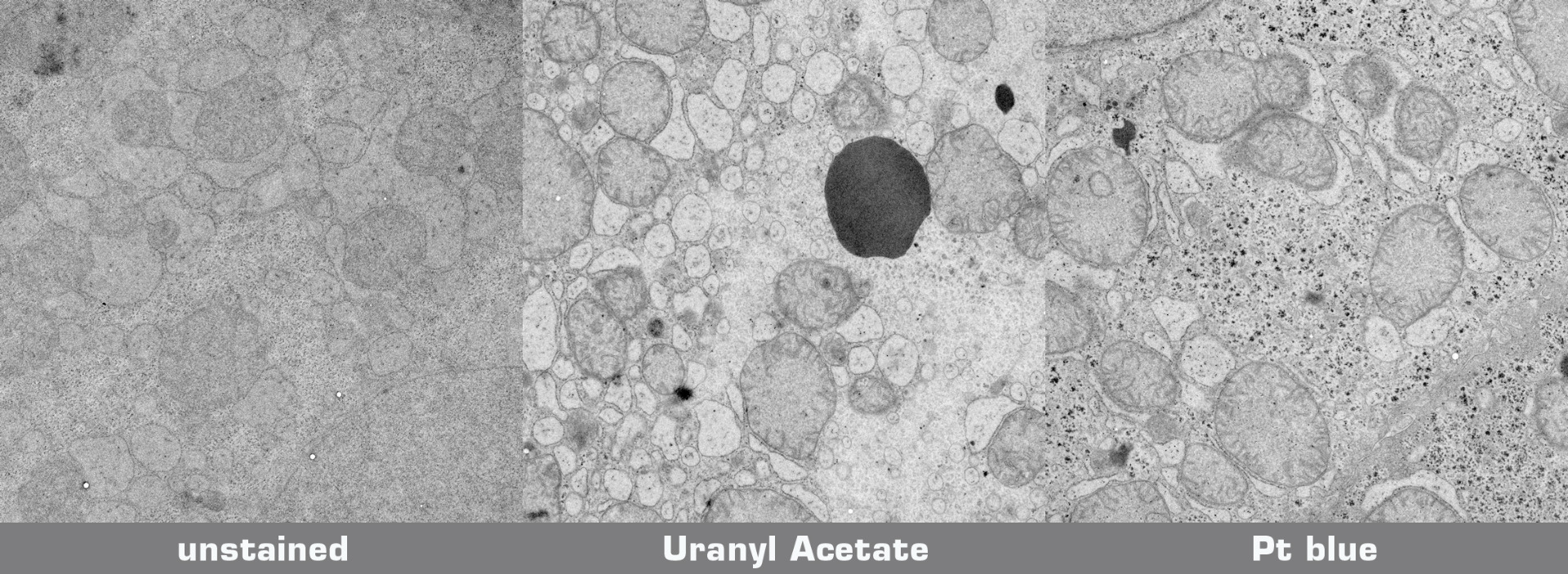
Testprotocol Pt blue vs. Uranyl Acetate
Mouse liver was chemically fixed (glutaraldehyde, osmium tetroxide), dehydrated and embedded in Epoxy resin without uranyl acetate en bloc staining. A Candida yeast culture was high pressure frozen, freeze substituted in 1% OsO4 in acetone, and embedded in Epoxy resin. 70 nm ultrathin sections mounted on Formvar coated grids were either stained with uranyl acetate/lead citrate orPlatinum blue/lead citrate:
* UA staining: Grids with sections were floated upside down on droplets of aqueous 2% UA while protected from light. Subsequently, samples were rinsed vigorously and then floated upside down on droplets of Reynold's lead citrate for 5 min in the presence of NaOH pellets. Finally, grids were washed vigorously and air dried before inspection in the EM. All steps were carried out at room temperature.
* Pt blue staining: Pt blue stock solution was diluted 1:100 in pH adjusted water (pH 3.5 with acetic acid). Grids with sections were floated upside down on droplets of the Pt blue dilution. Subsequently, samples were washed 5x2 min on ddH2O, dried completely, and then floated upside down on droplets of Reynold's lead citrate for 2 min in the presence of NaOH pellets. After lead citrate staining, grids were washed vigorously with a water stream from a pipette and air dried before inspection in the EM. All steps were carried out at room temperature.
Mouse liver:
Candida yeast culture:
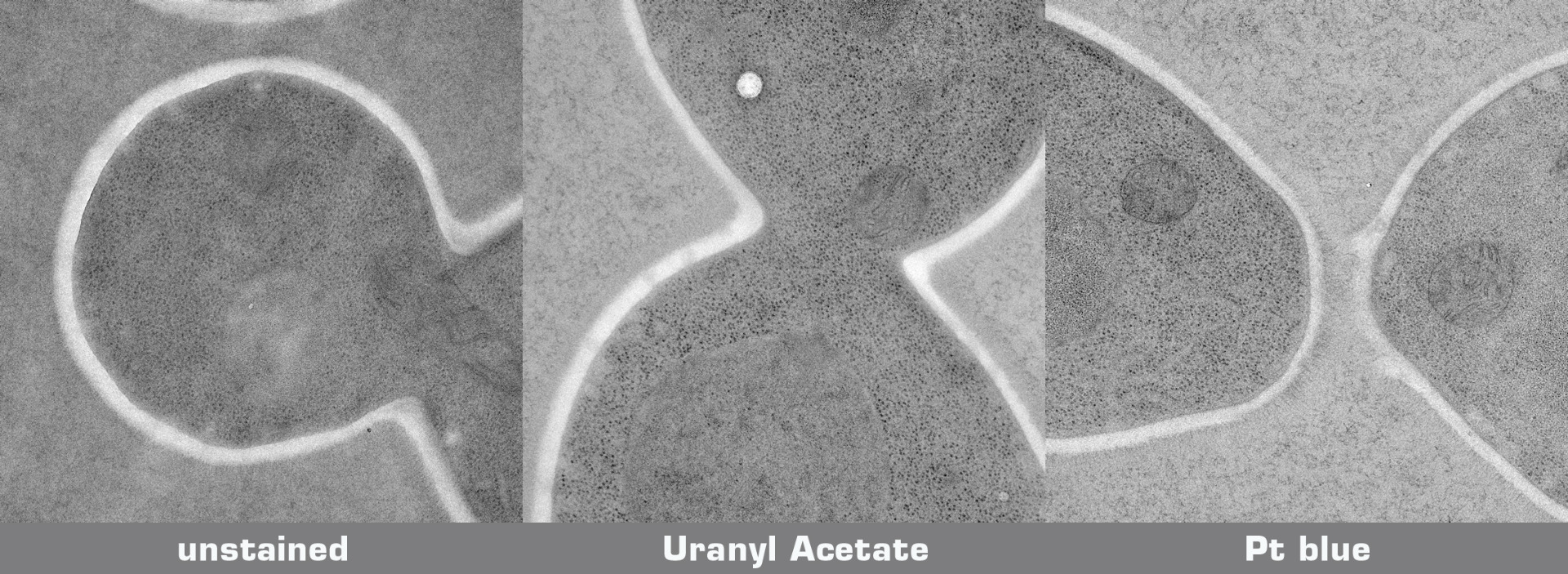
Poststained sections and unstained sections for comparison were imaged in a transmission electron microscope at 200 kV on a 4k CCD camera using comparable beam conditions. Digital images were optimised for contrast using the same settings.
Credits: Specimen preparation: Harald Kotisch, Vienna Biocenter Core Facilities GmbH; Staining and imaging: Guenter Resch, Nexperion e.U. - Solutions for Electron Microscopy



