A little Story of Cell Counting
Cell counting is a standard procedure routinely used in (cell culture) laboratories, e.g. to maintain healthy cell cultures or before freezing cells. For many experiments the exact number of cells has to be known in order to e.g. adjust the amount of chemicals required, but also for the consistency and reproducibility of experiments. Furthermore, it is important to know the accurate cell count for protocols like transfection, cell proliferation studies or qPCR (quantitative PCR). Simultaneously, viability can be determined.
Originally developed for the determination of the blood count, glass counting chambers (hemocytometers) are used to identify the number of blood cells in a sample. Meanwhile many other counting chambers are available to count bacteria, viruses or other pathogens (in the blood).
Ways to count cells
The cell count can be obtained either by counting the cells manually in a counting chamber such as the traditional hemocytometer or by using automated methods like flow cytometers, spectrophotometers, coulter counters or cell counters that use an image analysis software.
A. Manual cell counting
B. Automated cell counting
A. Manual cell counting
 1) Counting chamber: For manual counting, a cell suspension is transferred into a special counting chamber (e.g. hemocytometer). A counting chamber is a glass/plastic slide into which a grid of a defined size is etched. The cells in several areas of the grid are then counted under the light microscope and the number of cells is determined by extrapolation to the total volume. If special dyes (e.g. trypan blue, which only stains dead cells blue) are added, the viability of the cells (i.e. the ratio of living cells to the total number of cells) can also be determined. Glass chambers can be reused indefinitely but only one or two samples can be counted at the same time. Plastic counting chambers are disposable (interesting e.g. for infectious material) and have two or 4 sample counting areas in one slide.
1) Counting chamber: For manual counting, a cell suspension is transferred into a special counting chamber (e.g. hemocytometer). A counting chamber is a glass/plastic slide into which a grid of a defined size is etched. The cells in several areas of the grid are then counted under the light microscope and the number of cells is determined by extrapolation to the total volume. If special dyes (e.g. trypan blue, which only stains dead cells blue) are added, the viability of the cells (i.e. the ratio of living cells to the total number of cells) can also be determined. Glass chambers can be reused indefinitely but only one or two samples can be counted at the same time. Plastic counting chambers are disposable (interesting e.g. for infectious material) and have two or 4 sample counting areas in one slide.
Application: routine cell counting for maintenance.
2) Plaque assay/CFU (colony forming unit) count on nutrient medium: If youwant to determine which subgroups of cells should survive a certain treatment (e.g. bacteria resistant to a given antibiotic) by choosing the growth conditions (media composition, oxygen content, etc...), the plaque assay is suitable. For this purpose, a highly diluted cell suspension is spread evenly on solid growth medium in a Petri dish. The cell colonies that emerge after about 12-14 hours are then counted manually. Since only the surviving cells form a colony this test doesn´t give any indication about viability in the starter cells.
Application: drug screening.
| Advantages of manual cell counting | Disadvantages of manual cell counting |
| Relatively fast | Cells may need to be diluted (less accurate) |
| Inexpensive, sustainable (when glass chamber is being used) | Adherent cells must be detached and well suspended (possibly disrupting the experimental conditions, affecting viability) |
| Cell number and viability at the same time | 1-4 samples only per chamber |
| Cells can be seen (visual inspection) | Subjective errors possible |
interesting products
B. Automated cell counting
1) Coulter counter (volume measurement of cells): A Coulter counter is measuring the change in electrical resistance in a solution through the cells it contains. Apart from counting particles (not just cells), a Coulter counter can measure their size, as well. Additionally, this method is not as expensive as flow cytometers making it all in all the method of choice for cell cycle experiments.
Application: method of choice for cell cycle experiments.
2) Spectrophotometry: Measures the absorbance of light/optical density (OD) in a cell suspension. The higher the cell concentration, the cloudier the cell suspension in which the cells grow. This method is used e.g. to check bacterial cultures for their growth.
Application: Growth monitoring in bacterial cultures.
3) Flow cytometry: In a flow cytometer cells in a solution flow individually through a laser beam causing a light scatter (or in case fluorescence labelled cells are used a fluorescence signal). A light detector collects and analyses the backscattered light/fluorescence thereby picking up not only the cell count but also differences in form, structure and color of the cells.
Application: sophisticated method for cell analyses.
 4) Image Analysis: Nowadays an increasingly large number of devices are available that count cells via an image analysis software. Many also have additional features that allow researchers to characterize cells further via the analysis of viability, cell size, cell cycle, fluorescence expression and apoptosis.
4) Image Analysis: Nowadays an increasingly large number of devices are available that count cells via an image analysis software. Many also have additional features that allow researchers to characterize cells further via the analysis of viability, cell size, cell cycle, fluorescence expression and apoptosis.
Many microscopes now are equipped with an AI-controlled imaging software that makes cell counting intuitive, fast and effective for the user (e.g. the AI cell counting module of the Labscope software for the Zeiss Axiovert, Primovert and Primo Star microscopes).
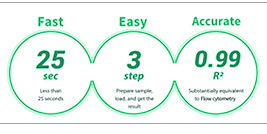
ADAMTM-MC2 is a fast and accurate automated fluorescent cell counter












